KCSE CHEMISTRY QUESTIONS AND ANSWERS PER TOPIC
|
The curves below represent the change in mass when equal masses of powered zinc and zinc granules were reacted with excess 2M hydrochloric acid. Study them and answer the question below.
Which curve represents the reaction with zinc granules? Explain your answer.
ANSWERS
X - Zinc granules
The gradient of the graph is less steep because there is less surface area.
0 Comments
The chromatogram below was obtained from a contaminated food sample P.
Contaminants Q, R, S and T are suspected to be in P. Use it to answer the following questions.
a) Identify the contaminants in mixture P.
b) Which is the most soluble contaminant in P.?
(a) State the Charles’ law
(b) A certain mass of gas occupies 146 dm3 at 291 K and 98.31 kPa. What will be its temperature if its volume is reduced to 133 dm3 at 101.325 kPa?
The scheme below shoes some reaction sequence starting with slid N. Study it and answer the questions that follows.
a) Write the formula of the complex ion in solution Q.
b) Write an equation for the reaction in step IV.
a) What would be observed if sulphur (IV) oxide is bubbled through acidified potassium manganate (VII)
(b) In an experiment, sulphur (IV) oxide was dissolved in water to form solution L. (i) What would be observed if a few drops of barium nitrate solution were immediately added to solution L? (ii) Write an ionic equation for the reaction that occurred between solution L and aqueous barium nitrate in (b) (i) above.
a) Write an equation for the reaction
b) Using the bond energies given below, calculate the enthalpy change for the reaction in (a) above
The table below gives the solubility’s of substances T and U at 10°C and 40°C.
When an aqueous mixture containing 55g of T and 12g of U at 80°c was cooled to 10°c, crystals formed.
a) Identify the crystals formed b) Determined the mass of the crystals formed c) Name the method used to obtain the crystals
Aqueous hydrogen chloride reacts with potassium manganate (VII) to produce chlorine gas, while a solution of hydrogen chloride in methylbenzene has no effect
on potassium manganate (VII). Explain this observation
Draw a labeled diagram to illustrate how alpha, beta and gamma radiations can be distinguished from each other.
When a sample of concentrated sulphuric acid was left in an open beaker in a room for two days, the volume was found to have increased slightly
Expected Answer
(a) Hygroscopy
(b) Drying of gases ║drying agent
When the oxide of element H was heated with powdered carbon the mixture glowed and carbon dioxide was formed. When the experiment was repeated using the oxide of element J, there was no apparent reaction.
Expected Answer
(a) Electrolysis of fused or molten oxide
(b) JCH║J, carbon, H
The information in the table below relates to elements in the same group of the periodic table. Study it and answer the question that follows:
Which element has the highest ionization energy? Give reason.
Expected Answer
G3, because it has the smallest atomic radius. Its outer most electron is tightly held by the nucleus or it requires a lot of energy to remove it.
A certain matchstick head contains potassium chlorate and sulphure. On striking the two substances react to produce sulphure dioxide and potassium chloride. Explain the environmental effect of using such matches in large numbers.
Expected Answer
SO2 which is poisonous is released in the air. Acid rain which may cause corrosion will be formed
Describe a simple laboratory experiment that can be sued to distinguish between sodium and sulphide and sodium carbonate.
Expected Answer
Add dilute acid (e.g. HCI or H2SO4) to each substances separately. If Na2S, colourless gas, smell of rotten eggs
Study the flowchart below and answer the questions that follow:
(a) Identify:
(i) X (ii) Y (b) State two uses of polyvinylchloride
(a) A student electroplated a spoon with copper metal .Write an equation for the process that took place at the cathode.
(b) Calculate the time in minutes required to deposit 1.184g of copper if a current of 2 amperes was used. (1 Faraday = 96500 coulombs, Cu=63.5).
The diagram below illustrates a method of preparing salts by direct synthesis
(a) This method can be used to prepare either aluminum chloride or iron (III) chloride.
Explain why it cannot be used to prepare sodium chloride. (b) Describe how a sample of sodium chloride can be prepared in the laboratory by direct synthesis.
ANSWERS
(a) It does not sublime.
(b) Cut a piece of Sodium metal, place it on a deflagrating spoon. heat it briefly then lower it into a gas jar of chlorine. It will continue burning forming Sodium Chloride.
In terms of structure and bonding, explain the following observations:
(a) The melting point of aluminum is higher than that of sodium (b) Melting point of chlorine is lower than that of sulphur
ANSWERS
(a) Aluminium has a stronger metallic bond because it has more delocalised electronsthan sodium.
(b) Sulphur has a ringed structure of S8 molecules whiles chlorine is diatomic. The forces in sulphur are stronger than chlorine.
Describe an experiment procedure that can be used to extract oil from nut seeds
ANSWERS
Write equations to show the effect of heat on each of the following:
(a) Sodium hydrogen carbonate (b) Silver nitrate (c) Anhydrous iron (II) sulphate
The set up below can be used to prepare oxygen gas. Study it and answer the questions that follow.
(a) Identify X
(b) What property of oxygen makes it possible for it to be collected as shown in the above set up? (c) State two uses of oxygen
a) State and explain what would happen if a dry blue litmus paper was dropped in a gas.
b) By using only dilute hydrochloric acid, describe how a student can distinguish between barium sulphite from barium sulphate. 10gm of sodium hydrogen carbonate were dissolved in 20cm3 of water in a boiling tube. Lemon juice was then added drop wise with shaking until there was no further observable change.
At 298K and 1 atmosphere, graphite changes into diamond according to the equation:
In the space provided, sketch a simple energy level diagram for the above change.
How would you obtain a sample of pure iodine from a mixture of iodine and lead sulphate?
Expected Response
Heat the mixture iodine sublimes and can be collected from the cool part of the test tube.
|
Chemistry Topics
All
Archives
December 2024
|
We Would Love to Have You Visit Soon! |
Hours24 HR Service
|
Telephone0728 450425
|
|
8-4-4 materialsLevels
Subjects
|
cbc materialsE.C.D.E
Lower Primary
Upper Primary
Lower Secondary
Upper Secondary
|
teacher support
Other Blogs
|



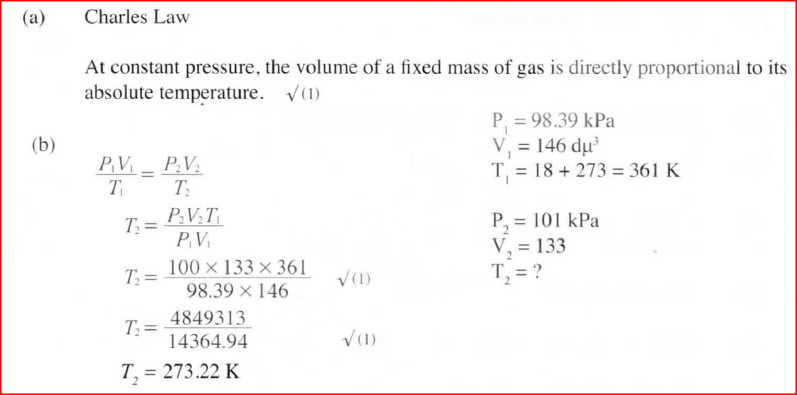
























 RSS Feed
RSS Feed