KCSE CHEMISTRY QUESTIONS AND ANSWERS PER TOPIC
|
(a) The diagram below shows the frasch process used for extraction of sulphur
Use it to answer the question that follows
(i) Identify X
(ii) Why is it necessary to use superheated water in this process (iii) State two physical properties of sulphur that makes it possible for it to be extracted by this method (b) The diagram below shows part of the process in the manufacture of sulphuric (VI) acid. Study it and answer the questions that follow
(I) Write an equation for the formation of sulphur (IV) oxide from sulphur
(II) What is the role of concentrated sulphur (VI) acid in chamber A? (III) Name two catalysts that can be used in the catalytic chamber B. (IV) State two roles of the heat exchanger (c) Explain one way in which sulphur (IV) oxide is a pollutant (d).what observation will be made when a few drops of concentrated sulphuric (VI) acid are added to crystals of sugar? Explain your answer
ANSWERS
(a)(i)Hot compressed air
(ii)To melt the sulphur and maintain it in molten state (iii)low melting point of sulphur - insolubility of sulphur in water - less dense than water (b)(i)S + O2 → SO2 (ii)To dry the SO2 and air (iii)Vanadium (v) oxide and platinum or titanium (iv)it provides the reactants (SO2 and O2) with enough energy to react - it removes heat from the product hence preventing decomposition or conserves heat, or recycles heat or reduces cost of production. (c)- contributes to acid rain which corrodes buildings OR - causes aquatic solutions to be acidic hence affecting aquatic life etc. - poisonous/toxic (d)Turns black conc H2SO4 removes hydrogen and oxygen from the sugar molecule leaving only carbon which is black . Dehydration of sugar forms carbon which is black.
0 Comments
The apparatus shown below shown below was used to investigate the effect of carbon monoxide on copper (II)oxide
a) State the observation that was made in the combustion tube at the end of the experiment.
b) Write an equation for the reaction that took place in the combustion tube c) Why is it necessary to burn the gas coming out of tube K?
The grid given below represents part of the periodic table. Study it and answer the questions that follow. The letters do not represent the actual symbol of the element
(i) Select a letter which represents an element that looses electrons most readily. Give a reason for your answer.
(ii) Explain why the atomic radius of P is found to be smaller than that of N (iii) Element M reacts with water at room temperature to produce 0.2 dm3 of gas. Determine the mass of M which was reacted with water. (molar gas volume at room temperature is 24 dm3 , relative atomic mass of M=7 (b) Use the information in the table below to answer the question that follows. (The letters are not the symbols of the elements)
Identify a letter which represents an element in the table that could be calcium, carbon or sculpture. Give reasons in each case.
(i) Calcium: Reason (ii) Carbon Reason (iii)Sulphur: Reason
ANSWERS
(a)(i)R - it has the largest atomic radius with the weakest nuclear attraction for outermost electron
(ii)Across the period the atomic radius decreases due to the increase in nuclear attraction Number of electrons in P is greater than in H
(b)(i) W - forms a basic oxide which forms an ionic bond
(ii)Y - the oxide is gaseous that forms a neutral solution (iii)U - the oxide is solid at room temperature, which is acidic with covalent bond
State and explain what would happen if a dry red litmus paper was dropped in a gas jar of dry chlorine.
ANSWERS
A solution was made by dissolving 8.2g of calcium nitrate to give 2 litres of solution. (Ca= 40.0; N=14.0; O= 16.0)
Determine the concentration of nitrate ions in moles per litre.
Explain why burning magnesium continues to burn a gas was bubbled
The burning magnesium produces more heat energy that the burning splint. The heat energy from magnesium is enough to break the sulphur oxygen bond setting free oxygen. Magnesium uses freed oxygen to continue burning.
Explain why a burning magnesium ribbon continues to burn when placed in a gas jar containing carbon (iv) oxide gas but a burning splint is extinguished.
(b) Write an equation for the reaction that takes place in (a) above.
a)what observation would be made if hydrogen sulphide gas was bubbled through a solution of zinc nitrate?
b) write an equation for the reaction that takes place in (a) above
In the presence of U.V light, ethane gas undergoes substitution reaction with chlorine.
(a) What is meant by the term? Substitution reaction: (b) Give the structural formula and the name of the organic product formed when equal volumes of ethane and chlorine react together.
When 15 cm3 of a gaseous hydrocarbon, P, was burnt in 100cm3 of oxygen, the resulting gaseous mixture occupied 70cm3 at room temperature and pressure.When the gaseous mixture was passed through potassium hydroxide solution, its volume decreased to 25cm3
a) What volume of oxygen was used during the reaction? b) Determine the molecular formula of the hydrocarbon
On heating a pale green solid K, carbon (IV) oxide gas and a black solid M were formed. On reacting K with dilute hydrochloric acid, carbon (IV) oxide gas and
green solution S were formed. When excess aqueous ammonia was added to solution S, a deep blue solution was formed. a) Identify the cation in solid K b) Identify the two onions in solution S.
When fuels burn in the internal combustion engine at high temperature, one of the products formed is nitrogen (II) oxide.
a) Write the equation for the formation of nitrogen (II) oxide. b) Give a reason why nitrogen (II) oxide is not formed at room temperature. c) Describe how formation of nitrogen (II) oxide in the internal combustion engine leads to gaseous pollution.
(a) State the Charles’ law
(b) A certain mass of gas occupies 146 dm3 at 291 K and 98.31 kPa. What will be its temperature if its volume is reduced to 133 dm3 at 101.325 kPa?
The scheme below shoes some reaction sequence starting with slid N. Study it and answer the questions that follows.
a) Write the formula of the complex ion in solution Q.
b) Write an equation for the reaction in step IV.
a) What would be observed if sulphur (IV) oxide is bubbled through acidified potassium manganate (VII)
(b) In an experiment, sulphur (IV) oxide was dissolved in water to form solution L. (i) What would be observed if a few drops of barium nitrate solution were immediately added to solution L? (ii) Write an ionic equation for the reaction that occurred between solution L and aqueous barium nitrate in (b) (i) above.
When a sample of concentrated sulphuric acid was left in an open beaker in a room for two days, the volume was found to have increased slightly
Expected Answer
(a) Hygroscopy
(b) Drying of gases ║drying agent
When the oxide of element H was heated with powdered carbon the mixture glowed and carbon dioxide was formed. When the experiment was repeated using the oxide of element J, there was no apparent reaction.
Expected Answer
(a) Electrolysis of fused or molten oxide
(b) JCH║J, carbon, H
A certain matchstick head contains potassium chlorate and sulphure. On striking the two substances react to produce sulphure dioxide and potassium chloride. Explain the environmental effect of using such matches in large numbers.
Expected Answer
SO2 which is poisonous is released in the air. Acid rain which may cause corrosion will be formed
Study the flowchart below and answer the questions that follow:
(a) Identify:
(i) X (ii) Y (b) State two uses of polyvinylchloride
a) The diagram below is part of set up used to prepare and collect dry chlorine gas.
i) Complete the diagram to show how a dry sample of chlorine gas can be collected.
ii) Name another substance and condition that can be used instead of manganese (VI) oxide. iii) Write an equation for each of the following; I. chlorine gas reacting with iron II. chlorine gas reacting with hot concentrated sodium hydroxide solution. b) An oxide of chlorine of mass 1.83g was found to contain 1.12g of oxygen. Determine the empirical formula of the oxide (O = 16.0; Cl = 35.5). c) Other than the manufacture of weed killers, name two other uses of chlorine.
In the laboratory, small quantities of nitric (V) acid can be generated using the following set up. Study it and answer the questions that follow.
a) i) Give the name of substance R.
ii) Name one other substance that can be used in place of sodium nitrate. iii) What is the purpose of using tap water in the set up above? b) Explain the following; i) It is not advisable to use a stopper made of rubber in the set-up ii) the reaction between copper metal with 50% nitric (V) acid in an open test-tube produces brown fumes. c) i) Nitrogen is one of the reactants used in the production of ammonia, name two sources of the other reactant. ii) A factory uses nitric (V) acid and ammonia gas in the preparation of a fertilizer. If the daily production of the fertilizer is 4800kg; calculate the mass of ammonia gas used in kg. (N = 14.0; O = 16.0; H = 1.0) iii) State two other uses of nitric (V) acid other than the production of fertilizers.
ANSWERS
(a) (I) Concentrated /sulphuric (VI) acid.
(ii) Potassium nitrate (iii) To condense the fumes or vapour of nitric (V) acid into liquid (b) (i) Nitric acid (V) will corrode the rubber (ii) The reaction produces nitrogen monoxide (colourless) which is oxidised by oxygen from the air to form nitrogen(IV) oxide. (c)(i) Water Alkanes Biogas Water gas
a) State and explain what would happen if a dry blue litmus paper was dropped in a gas.
b) By using only dilute hydrochloric acid, describe how a student can distinguish between barium sulphite from barium sulphate. A weighed sample of crystalline sodium carbonate (Na2CO3. H2O) was heated in a crucible until there was no further change in mass. Calculate the number of moles (n) of the water of crystallization
Describe how a solid sample of potassium sulphate can be prepared starting with 200cm3 of 2M potassium hydroxide.
Describe how the percentage by mass of copper in copper carbonate can be determined.
Draw and name the isomers of pentane.
|
Chemistry Topics
All
Archives
December 2024
|
We Would Love to Have You Visit Soon! |
Hours24 HR Service
|
Telephone0728 450425
|
|
8-4-4 materialsLevels
Subjects
|
cbc materialsE.C.D.E
Lower Primary
Upper Primary
Lower Secondary
Upper Secondary
|
teacher support
Other Blogs
|
















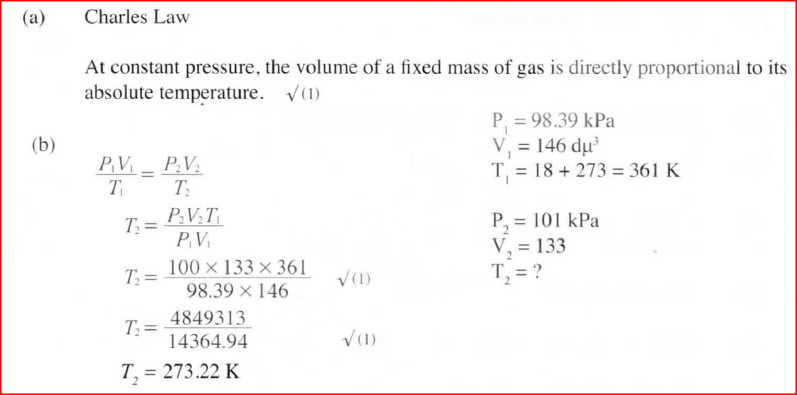

















 RSS Feed
RSS Feed